An alternative to antibiotics for CF patients
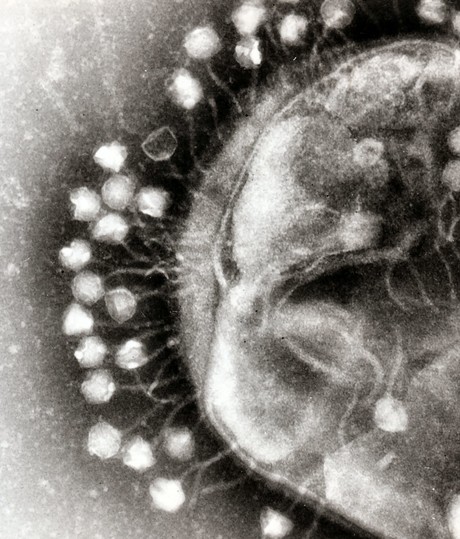
UK scientists have shown that bacteriophage (phage) therapy could offer a safe and effective alternative to antibiotics in the treatment of cystic fibrosis (CF) lung infections. Their study has been published in the journal Thorax.
Chronic lung infections caused by the bacterium Pseudomonas aeruginosa are becoming increasingly difficult to treat due to antimicrobial resistance. With limited alternative therapeutic options available, this has led to renewed interest in the use of phages — viruses that infect and kill bacteria but are otherwise harmless.
A major advantage of phages is that they only target the harmful bacteria, so there are less of the side effects often associated with antibiotics. Unfortunately, phage therapy has not had the same level of funding as drug development due to a lack of convincing preclinical efficacy studies.
Now, researchers led by the University of Liverpool have shown that phage therapy is highly effective in treating established and recalcitrant chronic respiratory tract infections caused by multidrug-resistant P. aeruginosa strains. Significantly, they show that phages are capable of killing the bacteria in long-term infected lungs, such as those suffered by patients with cystic fibrosis.
“Cystic fibrosis patients face the prospect of life-long treatment with antibiotics, which often prove ineffective and can have side effects, especially when used for long periods,” said Professor Craig Winstanley, who co-led the study. “Hence, phage therapy could be a particularly valuable addition to the treatment of chronic lung infections in these patients.”
The WHO recently identified P. aeruginosa as one of the key pathogens against which there is a critical need to develop new therapies. In light of this, the study provides valuable preclinical evidence for phage therapy being a viable option.
Lipid science receives new Australia-led online hub
The Lipid Network launched this month to build links between researchers, clinicians and industry...
Proposed Australian trial eyes up blood testing for Alzheimer's
Utilising networks of GPs across three states, a large-scale study intends on combining digital...
Applications open for $300K pandemic therapeutics fellowships
Applications are now open for the Cumming Global Centre for Pandemic Therapeutics' inaugural...




