ATAGI recommends AstraZeneca for Sydneysiders
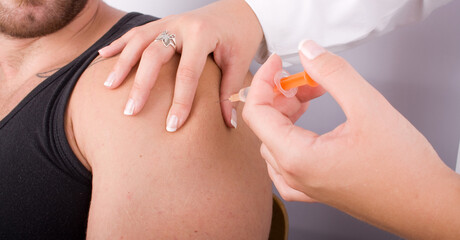
The Australian Technical Advisory Group on Immunisation (ATAGI) has once again revised its COVID-19 vaccine advice, stating that all individuals aged 18 years and above in Greater Sydney should strongly consider getting vaccinated with any available vaccine — including COVID-19 Vaccine AstraZeneca. This is on the basis of the increasing risk of COVID-19 and ongoing constraints of Pfizer supplies.
ATAGI has indicated that as the COVID-19 outbreak in NSW continues to grow, the risk of disease — particularly in the Greater Sydney area — is likely to continue to be significant over coming weeks. The Group has previously advised that the benefits of the AstraZeneca vaccine are greater in large outbreak situations than the risk of rare side effects for all age groups. All adults in Greater Sydney should therefore strongly consider the benefits of earlier protection with AstraZeneca rather than waiting for alternative vaccines.
A second reason for ATAGI to recommend vaccination at this time is emerging data about severity of disease, with the Delta variant appearing to be more severe than the original SARS-CoV-2 strain. The proportion of people less than 60 years requiring hospitalisation appears to be higher than was reported in outbreaks with the original SARS-CoV-2 strain, which reinforces the benefit of protection with any available vaccine. Maximal protection requires two doses of vaccine, but even a single dose of either vaccine provides >70% protection against hospitalisation. Furthermore, a single dose of AstraZeneca vaccine reduces risk of transmission by around half.
ATAGI has previously issued advice recommending a shorter interval between the first and second doses of AstraZeneca vaccine of four to eight weeks in an outbreak (versus the routine 12-week interval) so that maximal protection against COVID-19 can be achieved earlier. In addition to this, the Group has suggested increasing the interval between the first and second doses of Pfizer vaccine from three to six weeks, which would allow limited vaccine supplies to be redirected to obtain first-dose protection in outbreak areas of greatest need.
Please follow us and share on Twitter and Facebook. You can also subscribe for FREE to our weekly newsletters and bimonthly magazine.
MV Hondius passengers return to Australia under strict infection control measures
The Australian CDC has provided an update on hantavirus risk to Australians and infection...
$96m RNA Research and Manufacturing Facility opens in NSW
The new 4500 sqm RNA Research and Manufacturing Facility within the Macquarie University...
Cartherics and Catalent announce enhanced partnership
To support immune cell therapy products for the treatment of cancer and endometriosis, Australian...




