TGA approves first treatment for geographic atrophy
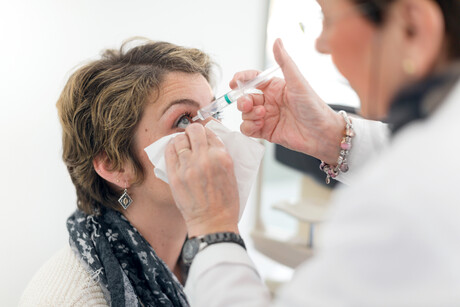
The Therapeutic Goods Administration (TGA) has approved the use of the first treatment in Australia for geographic atrophy (GA) — a disease which causes severe, progressive central vision loss and is a leading cause of legal blindness for people over 50. The approval has been welcomed by the macular disease community, who to date have had limited treatment options.
More than 75,000 Australians are living with GA, also known as late ‘dry’ age-related macular degeneration (AMD). It is a progressive and irreversible disease caused by the growth of lesions, which destroy the retinal cells responsible for vision. The vision loss caused by GA severely impairs independence and quality of life.
Australia has now become the first country outside of the United States to approve the use of the drug pegcetacoplan (SYFOVRE) to slow down the progression of GA. The approval is based on results from the Phase 3 OAKS and DERBY studies at 24 months, which demonstrated that regular eye injections with pegcetacoplan slowed down lesion growth.
“For the first time, Australians with GA will have a treatment to slow the progression of this irreversible form of vision loss, said Dr Jeffrey Eisele, Chief Development Officer at Apellis Pharmaceuticals. “Building on the success in the US, we are excited to bring SYFOVRE to even more patients who are impacted by this devastating disease.”
Professor Robyn Guymer AM led the Centre for Eye Research Australia’s (CERA) site of the international clinical trials into the new drug and was on the global advisory committee for Apellis. She said the approval of a treatment that can slow the progression of GA is a positive first step, with scientists now hoping to continue research which aims to find ways of preventing damage before it occurs.
“As a retina specialist, I have seen how GA often takes away a person’s ability to read, drive and even see faces of their loved ones,” Guymer said. “The approval of SYFOVRE is a historic moment full of hope for the Australian GA community, who have been waiting for a treatment.”
Dr Kathy Chapman, CEO of Macular Disease Foundation Australia, said the approval of SYFOVRE is welcome news but that not everyone with GA will necessarily be eligible for the treatment. Those with GA should therefore consult their ophthalmologist, who will weigh up the benefits and risks to determine if the new treatment is suitable for them.
AusBiotech partners with Tenmile
Designed to support Australia's homegrown life sciences innovation, AusBiotech has announced...
Australian CDC issues update in wake of Ebola outbreak
After the WHO determined the outbreak of Ebola in the DRC and Uganda to be a public health...
Australia announces $7.2m diphtheria outbreak response package
To respond to the biggest diphtheria outbreak on record, support has been announced for the NT...




