Thermostable vaccine can be stored at warm temperatures
A new type of vaccine that can be stored at warmer temperatures, removing the need for refrigeration, has been developed for the mosquito-borne virus Chikungunya. The vaccine candidate has been engineered using a synthetic protein scaffold that could revolutionise the way vaccines are designed, produced and stored, with the results published in the journal Science Advances.
Infectious diseases continue to plague populations worldwide — particularly in the form of epidemics caused by Ebola, Zika and others — with developing countries often bearing the brunt of such outbreaks as they lack adequate infrastructure and resources. A recent example is Chikungunya, a virus transmitted by the bite of an infected mosquito that causes crippling headache, vomiting, swelling of limbs and potentially death. Even if a fever ends abruptly, chronic symptoms such as intense joint pain, insomnia and extreme prostration remain.
Formerly confined to sub-Saharan Africa, Chikungunya has recently spread worldwide as its mosquito host leaves its natural habitat due to deforestation and climate change, with recent outbreaks in USA and Europe causing alarm. Now, researchers from the University of Bristol and the French National Centre for Scientific Research (CNRS) have teamed up with computer technology company Oracle to find a way to make vaccines that are thermostable (able to withstand warm temperatures), can be designed quickly and are easily produced.
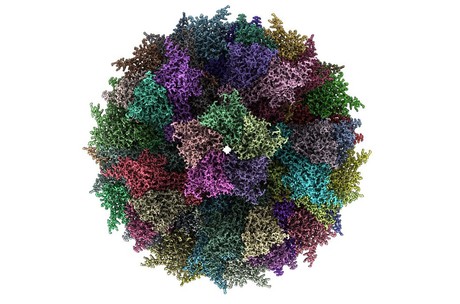
“We were working with a protein that forms a multimeric particle resembling a virus but is completely safe, because it has no genetic material inside,” said Pascal Fender, expert virologist at CNRS. “Completely by chance, we discovered that this particle was incredibly stable, even after months without refrigeration.”
“This particle has a very flexible, exposed surface that can be easily engineered,” added Imre Berger, Director of the Max Planck-Bristol Centre for Minimal Biology in Bristol. “We figured that we could insert small, harmless bits of Chikungunya to generate a virus-like mimic we could potentially use as a vaccine.”
To validate their design, the scientists employed cryo-electron microscopy, a powerful new technique recently installed in Bristol’s state-of-the-art microscopy facility. Cryo-EM yields very large datasets from which the structure of a sample can be determined at near atomic resolution, requiring massive parallel computing.
Enabled by Oracle’s high-performance cloud infrastructure, the team developed a novel computational approach to create an accurate digital model of the synthetic vaccine, with University of Bristol IT specialists Christopher Woods and Matt Williams, together with colleagues at Oracle, implementing software packages on the cloud. Woods explained: “We were able to process the large datasets obtained by the microscope on the cloud in a fraction of the time and at much lower cost than previously thought possible.”
“Researchers have had a long tradition of building and installing their own supercomputers on-premises, but cloud computing is allowing them to run large datasets in record time, with fast connectivity and low latency,” added Phil Bates, leading cloud architect at Oracle. “This is helping them crunch data and make scientific breakthroughs much faster.”
The particles the scientists designed have yielded promising results in animal studies, setting the stage for a future vaccine to combat Chikungunya disease. As noted by Berger, “Our vaccine candidate is easy to manufacture, extremely stable and elicits a powerful immune response. It can be stored and transported without refrigeration to countries and patients where it is most needed. Intriguingly, we can now rapidly engineer similar vaccines to combat many other infectious diseases just as well.”
“It really ticks a lot of boxes,” added Fred Garzoni, founder of Bristol biotech start-up Imophoron, which is developing new vaccines derived from the present work. “Many challenges in the industry require innovative solutions, to bring powerful new vaccines to patients. Matching cutting-edge synthetic biology with cloud computing turned out to be a winner.”
Please follow us and share on Twitter and Facebook. You can also subscribe for FREE to our weekly newsletters and bimonthly magazine.
How an Alzheimer's-linked protein shapes long-term memories
Tau, a protein widely associated with memory loss in dementia, has been shown to be essential for...
Multiple myeloma treatment reprograms patients' own immune cells
An Australian and Victorian Government funding agreement has allowed eligible Australians with...
Could 'fusion proteins' reduce errors in a promising gene-editing tool?
Gene-editing tool CRISPR-Cas9 is used, among many things, to engineer new cancer immunotherapies....




