Redesigning and refurbishing laboratories in hospitals
Wednesday, 05 May, 2010
The Centre for Transplant and Renal Research (CTRR) laboratories of the Westmead Millennium Institute for Medical Research (WMI) at the Westmead Hospital campus have been redesigned and refurbished. Gary Martinic used his significant experience with laboratory and animal facility design and refurbishment to centralise four separate laboratories into one in just eight weeks. This article should serve as a useful guide for those new to the laboratory refurbishment process.
Westmead Hospital is part of the greater Sydney West Area Health Service. It is a 975-bed hospital, and one of which services the population of 1.5 million of the south-west Sydney region. The WMI is located on the same campus as Westmead Hospital, Westmead Children’s Hospital and the Children’s Medical Research Institute.
As a medical research organisation, the WMI has approximately 237 wet lab and 110 dry lab researchers and 52 administrative and support staff. The research facilities of all of these organisations comprise the Westmead Research Hub, which provides a cooperative environment in which major equipment and scientific expertise can be shared for a more cost-effective and efficient approach to medical research. There are 900 researchers in the hub (including WMI staff).
|
|
Rationale for the CTRR laboratory refurbishment
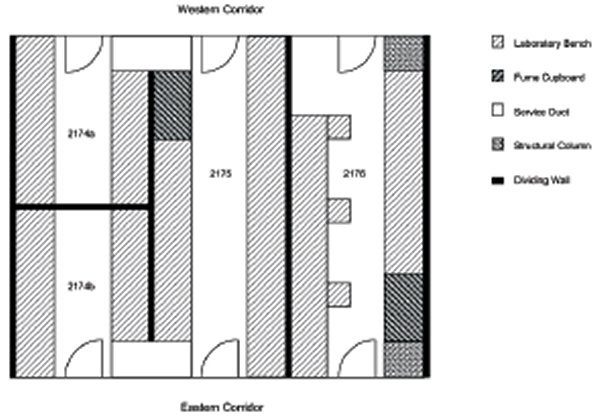
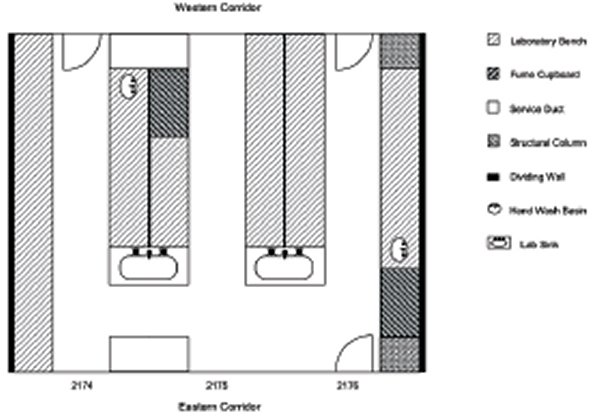
The reasons for the refurbishment of the CTRR laboratory were many; each individual laboratory was isolated by being ‘closed-off’ from the next, thereby not permitting daily interaction of lab staff (Figure 2). Also, the individual labs were far too small, poorly serviced and ventilated, did not fit the workflow of day-to-day laboratory work and reduced essential seating capacity due to poorly designed bench configuration (Figure 3).
|
|
|
|
These labs were also ‘land-locked’ within the CS corridor and had no access to natural light, so incandescent fluorescent lighting remained the only viable alternative.
At least initially, we proposed that a large proportion of laboratory space would be dedicated towards the construction of a ‘sterile suite’ in which to expand the tissue culture and sterile work procedures area (which included PCR and cell separation work); however, as all tissue culture work was transferred to our primary cell culture laboratories, located in another region of the CS corridor, this requirement was no longer necessary. We did need to divide the proposed new laboratory into a ‘clean area’ for work with PCR and microarrays and into a more ‘generic’ work area.
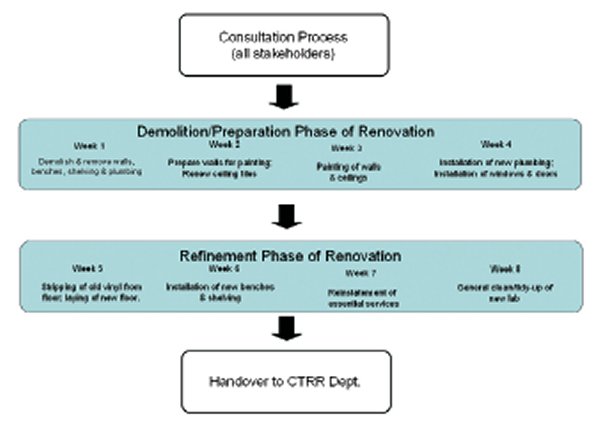
Consultation process
It is important that any envisaged project be planned thoroughly before refurbishment work commences. Naturally, the end result is better achieved if the laboratory manager (LM) and project manager(s), in the first instance, discuss the requirements of what is needed with the end users of the facility (the scientists), regarding the type of work they are doing in the facility, as this will affect the outcome of the refurbishment.
As the management representative of the laboratory, the LM needed to meet with and discuss the refurbishment requirements with the research heads of department to confirm what they would like to see achieved.
Once confirmed, the LM met with the organisational executive to outline the proposed plans and to obtain agreement to fund the project.
Following this, the LM prepared a plan of alternative options for the construction project manager and his team, starting from the least expensive to the most expensive option. This was displayed by comparing the actual and proposed layout, or floor plan, of the laboratory.
Significant discussion then took place between the LM and project manager to determine the most optimal, timely and cost-effective way in which the refurbishment outcome could be achieved. The project manager then priced the intended work and provided a written quotation. Upon reviewing the itemised quotation, the LM discussed the quotation with the heads of department and the organisational executive and, upon receiving their agreement, confirmed with the project manager that the refurbishment could commence.
Methods - CTRR refurbishment overview
The CTRR had two existing laboratories (dimensions 10.5 x 3.5 m) and two smaller partitioned laboratories (5.25 x 1.75 m) located on level 2 of the CS corridor, D block of WH (Figure 3 shows laboratory bench configuration pre-refurbishment).
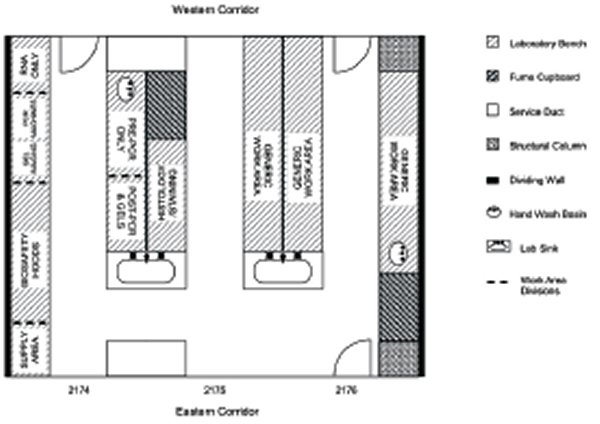
We wanted these labs to be opened up into one larger centralised laboratory, thus we considered how best to redesign the rooms to maximise functionality by improving workflow and to consolidate departmental resources. We felt that the optimum bench configuration was best achieved by shortening the length of the benches at the eastern corridor end, to allow ‘walk-around’ capability (Figure 4).
|
|
The bench configuration would need to provide generic areas for general laboratory work as well as designated areas for more specialised work, eg, gel electrophoresis, molecular biology and pre- and post-PCR work (Figure 5).
|
|
We considered four different design options, which included:
- Option One: Leave laboratory as is but install interlinking doorways at eastern end of lab only. In effect, dividing walls between rooms would remain intact, although walk-through access between rooms would be achieved by the installation of doorways at the eastern end of the CS corridor. This option retained existing lab shelving for chemical storage.
- Option Two: Remove stud walls above the electrical race region (which incorporates a horizontal services duct to reticulate power, gas and air), thus remove upper portion of walls from above bench to ceiling.
- Option Three: Remove stud walls completely (ceiling to floor).
- Option Four: Completely dismantle and dispose of dividing stud walls, all existing fixtures and joinery. Appoint architects to redesign and equip laboratory with a new modular lab furnishing throughout.
Other modifications common to options 1-3 included:
- Dismantle and remove old benches and install new benches (8.9 metres long and 600 mm wide installed either side) but shorten length of benches at eastern corridor end to allow ‘walk-around’ capability of 1.6 m.
- Install new chemical reagent shelving as dividers on new benches (except option 1).
- Reinstate all essential services (power, water, gas, air, oxygen) to new joinery.
- Purchase and install new under-bench cabinets.
Regulatory requirements
Before any refurbishment work could commence, the LM had to write to the Secretary of the local Institutional Biosafety Committee (IBC) seeking permission from the Office of the Gene Technology Regulator (OGTR) in Canberra to structurally modify the CTRR laboratory, as only three (Rms. 2174a, 2174b and 2175) of its four rooms were certified to PC2 (physical containment level 2) status, but one (Rm.2176) was not.
The Gene Technology Amendment Act 2007 requires that “prior to any significant structural changes that will affect the containment of Genetically-Modified Organisms in the facility, the applicant must request a suspension of certification, in writing from the Regulator”. Thus, PC2 certification status was suspended during the refurbishment period. As the entire refurbished facility was to be certified as PC2, communications concerning the new ‘envelope’ were made to the OGTR and, following inspection by the local IBC, a variation of certification was submitted to the OGTR. On completion of refurbishment work, and consequent inspection by IBC, PC2 certification was re-instated.
One of the most important issues to clarify is to distinguish between wet labs and dry labs, and core facilities. Of course, one of the reasons for defining wet labs and dry labs is cost. Wet labs are considerably more expensive than dry labs and so flexibility of wet lab use is critical. Both can be built on the same floor (of a multi-storey building) but the two should be kept in separate areas.
Dry labs may include anything that is essentially ‘non wet lab’; for example, this may include bio-informatics, gene sequencing, clinical trials facilities. Core facilities usually include shared high technology platforms which may include electron microscopy, flow cytometry, DNA microarray, histology services, etc, and so adaptability is important in such shared-resource areas.
The new CTRR laboratory would remain primarily a wet lab, including RNA, pre- and post-PCR work areas, gel-electrophoresis and imaging facilities, located in the ‘clean area’ along the southern wall (Figure 5, former labs 2174a/b) of the new laboratory.
While all of the refurbishment options required some degree of removal of dividing walls (Figure 6), option two improved lab functionality in the most cost-effective way. Damaged ceiling tiles were renewed and the entire suspended ceiling was spray-painted with three coats of off-white paint to give a modern finish, after which brighter incandescent lighting was installed (Figure 7).
|
|
|
|
Large viewing windows were installed at both sides of the eastern and western corridors to encourage both visibility and interaction (Figure 7).
The existing vinyl floor was removed and was stripped down to bare concrete. The next day, sheet vinyl flooring (Tarkett Safety Flooring, Sydney) which is chemical/stain/heat resistant and non-slip, was installed (Figure 8), with joins being seam-welded. Total floor area covered approx. 140 m2, and included coved edging 150 mm high.
|
|
Existing bench joinery was removed and discarded, as a decision was made to install new chemical reagent shelving (Figure 9) and lab benches (Figure 10), and to purchase new, mobile under-bench cabinets which were later delivered and installed by the supplier. All walls and doors were also spray-painted. Existing air-conditioning services to the laboratory were working efficiently and so were left intact; however, the airflow to the new laboratory had to be re-balanced to control airflow issues in close proximity to the installed Class II biosafety airflow cabinets.
|
|
|
|
All essential services (electricity, water, gas, air, medical oxygen) to the laboratory were also left intact as much as possible in order to keep costs down. To avoid impact on laboratory areas, and to minimise costs, chemical fume cupboards were left in place.
In order to create one centralised laboratory from the existing dividing-walled areas, we blocked off three out of six original entry doors (western and eastern corridors (see Figure 2) which had three existing doors on either side). This change meant that two entry doors were left on the western corridor side and one door on the eastern corridor end (Figures 4, 5).
Option one was the least expensive; however, this option isolated the staff because of dividing walls, thus it reduced the chances of daily interaction (staff could not see each other across the lab, although walk-through doorways would be installed at the eastern end of the corridor). Left in this state, individual labs were disjointed and secluded from each other.
Option two was chosen because it allowed an open-plan, centralised (walk-through) laboratory at an inexpensive price, while also allowing most of the existing essential services to remain in place. This option permitted staff to easily see each other across the entire laboratory.
Option three was not chosen because it necessitated complete removal of stud walls, thus all existing essential services would need to be re-instated, which was not only expensive, it created unnecessary installation work.
Option four was not chosen, being the most expensive and the most time-consuming option.
Apart from the physical renovation of the laboratories, the operational functionality and the way the new laboratory was managed also changed. Well before any refurbishment work took place, all stakeholders met on numerous occasions to conceptualise a novel work approach for the new laboratory which would see it used by the bench scientists as an open-plan, multiple-user, shared-resource, ‘round-the-clock facility’. A booking system was implemented for each large laboratory instrument and scientists were obliged to make a booking in order to use equipment well in advance of intended use.
Also, we placed less emphasis on ownership of bench space, and more emphasis on working cohesively towards common research goals in the new laboratory. While this untraditional approach to day-to-day work management made some staff feel like ‘nomads’ and it did take time for some staff to adapt, the outcome was successful. We have found since that staff work to this approach very well, and we have managed to integrate three separate research groups working cohesively within the one centralised CTRR laboratory under this arrangement.
Needless to say, this ‘round-the-clock’ work approach is not an entirely novel concept as it has been in use by many laboratories in the United States and in parts of Europe; however, the concept was new to our department and it took time before it became a successful operational model. Also, it essentially came out of necessity, as the CTRR department was expanding, yet it was impeded due to ongoing spatial constraints.
Summary
The simple re-design (option two) of the CTRR laboratory, provided an open-plan laboratory, while leaving essential services and larger fixed laboratory machinery (Figure 11) mostly in place. It provided a centralised, easy-to-walk-through laboratory which could be easily viewed from both eastern and western corridors of CS, and which catered for both generic and specialised work zones, while also increasing essential seating capacity.
|
|
Unnecessary doorways were reduced by half, thus significantly reducing airflow balance and infection control issues.
Perhaps most importantly, this was achieved in a timely and cost-effective way, by utilising the experience of the LM and senior lab staff, without engaging the services of architectural firms. In the end, the original concept of creating a new laboratory which improved workflow and allowed a consolidation of CTRR resources was achieved.
*Gary Martinic is the Laboratory and Operations Management, Centre for Transplant and Renal Research and Centre for Infectious Diseases and Microbiology, Westmead Millennium Institute for Medical Research, Westmead NSW 2145 Australia.
The author would like to thank the following WMI and CS staff for their helpful comments and advice: Glenn Holden for general input and planning; Mark Smith for handling of regulatory PC2 practices and dealing with the office of the Gene Technology Regulator in Canberra; Professor David Harris (Kidney Failure Research Group) and Dr Wayne Hawthorne (Xeno-Islet Transplant Group) for their critical review of the manuscript; Gary Cole and ‘Cole Constructions’ Pty Ltd for their dedicated, coordinated and efficient efforts during this refurbishment period. Mark Dado, Chief Operating Officer, on behalf of the executive of the WMI, is also thanked for giving permission for this work to be published.
Scientific rigour within a person-centred, biophilic setting
The $96 million, 4500-square-metre RNA Research and Manufacturing Facility has opened within the...
Sydney's $490m commercial life sciences precinct gets first tenants
ION, a dedicated, purpose-built commercial life sciences precinct located in Waterloo, has...
Project PEARL: safety findings of a lab ventilation systems comparison
Could this independent comparison test of lab ventilation systems under real-world stress...