Molecular diagnostics and the fight against coronavirus

Emerging pathogens, such as Ebola, Zika, Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS), are becoming a growing threat due to urbanisation, climate change and the global migration of people. The current outbreak of a novel coronavirus (COVID-19) has been of particular concern, having spread to at least 25 nations since it was first encountered in China late last year.
COVID-19 has affected more than 42,000 people and cost more than 1000 lives at the point when this article is being processed. The global economic loss is expected to be over $40 billion, larger than that suffered during the SARS outbreak in 2003.
Early detection and diagnosis are essential to reduce the prevalence of infectious diseases and to prevent them from becoming epidemic or even pandemic. What are the methods available to detect infectious diseases? Figure 1 summarises the methods currently available.
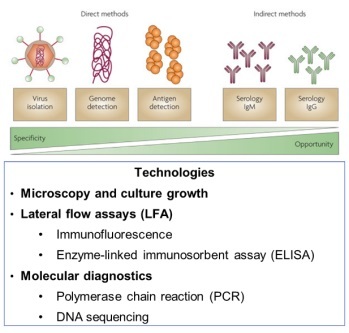
Traditionally, pathologists have used culture growth and microscopy. However, it usually takes several days to grow bacteria in medium and is not suitable for detecting viruses due to their small size and slow growth. In a fast-paced medical world, an immediate and accurate diagnosis and quicker methods are necessary. A faster and cheaper method would be lateral flow assays (LFA), such as immunoassays (using antibodies) or enzyme-linked immunosorbent assay (ELISA). Such a device is similar to a pregnancy test strip and does not require expensive equipment. However, this method hardly works for the new strain of coronavirus due to the limitation of non-specification, low sensitivity and long development process (up to three months).
For the rapid detection of COVID-19, however, molecular diagnostics (MDx) is the best, if not the only, option at the moment. MDx consists of diagnosis at the molecular level, identifying the DNA or RNA of a pathogen. This is a fast-changing field driven by two trends: DNA chips or PCR-enabled point-of-care MDx and next-generation DNA sequencing. In this article, IDTechEx discuss how the PCR technique is being used to tackle the coronavirus diagnosis. This article is drawn from ‘Molecular Diagnostics 2020–2030’, which offers the full landscape of molecular diagnostic market, analysis of players, assessment of innovations, emerging market and 10-year market forecast.
How molecular diagnostics is helping fight the spread of coronavirus virus
In December 2019, Chinese scientists identified the RNA sequence of COVID-19 by the whole genome sequencing method, classifying COVID-19 as a positive-sense RNA virus. Based on the sequence, the first coronavirus detection kit was developed by a group of researchers from the German Center for Infection Research (DZIF) at Charité hospital in Berlin.
This world’s first diagnostic test for the coronavirus is based on reverse transcription polymerase chain reaction (RT-PCR) and has now been made publicly available following its online publication by the World Health Organization (WHO). More than 52 companies have now developed analogous diagnostic kits within weeks in China. Seven of them have been approved by the National Medical Products Administration (NMPA) and received a medical device registration certificate, as listed in Figure 2. So far, the dominant method is real-time PT-PCR. Other methods based on DNA microarrays are under development.
| Companies | Method | Detection time | Sensitivity (copies/mL) |
Samplea | Target gene |
| Sansure Biotech | Real-time PCR | 2 hours for 96 samples or 30 min |
200 | OP swab or lung wash |
ORF1ba / N |
| Shanghai Geneodx Biotech |
Real-time PCR | About 2.5 hours | N/A | NP/OP swab | ORF1ba / N |
| Da An Gene | Real-time PCR | N/A | N/A | NP/OP swab | ORF1ba / N |
| Shanghai BioGerm Medical Biotechnology |
Real-time PCR | N/A | N/A | NP/OP swab | ORF1ba / N |
| Liferiver | Real-time PCR | N/A | 1000 | NP/OP swab or lung wash |
ORF1ba / N / E |
| BGI groupb | Real-time PCR | N/A | N/A | NP/OP swab or lung wash |
ORF1ba |
| a: NP/OP swab: Nasopharyngeal swab and oropharyngeal swab. b: BGI also obtained an approval for its data analysis systems. |
|||||
Before using the MDx kit to detect COVID-19, the sample needed to be treated. Once collected, the sample is then transferred to a central lab where technical staff will extract and purify the RNA before adding to the COVID-19 diagnostic kit. The suppliers of the reagents and systems for the extraction and purification include Thermo Fisher, Qiagen, bioMérieux and Roche. Many of these companies are providing ready-to-use RT-PCR workflow products to tackle the COVID-19 outbreak.
It is a still ongoing battle to fight against COVID-19 and the public are increasingly aware of the big threat of the spread of infectious diseases. As traditional diagnosis fails to cope with novel pathogens and transmission of pathogens becomes faster, molecular diagnostics will rise to sit at the core of prevention of pandemic tragedies. Indeed, MDx is one of the fastest-growing markets in the healthcare industry, driven by the fast-developing technologies such as the third-generation DNA sequencing, digital PCR and microfluidic devices. IDTechEx forecasts MDx for infectious diseases detection will become a US$7 billion market by 2030.

‘Molecular Diagnostics 2020–2030’ covers the technologies, benchmarking and players of DNA arrays, PCR devices and DNA sequencing from the hardware. The report highlights the recent innovations and trends in digital PCR, third-generation DNA sequencing and more. It also provides a 10-year forecast on each technology segment and end-user market including infectious disease, cancer, liquid biopsy, genetic testing, companion diagnostics and beyond.
Please follow us and share on Twitter and Facebook. You can also subscribe for FREE to our weekly newsletters and bimonthly magazine.
Protein build-up in brain blood vessels and dementia risk
In the US, health information of over 1.9 million adults aged 65 and older has led to suggested...
Proteomic approach identifies new blood-clotting disorder
Analysis of lethal antibodies has helped Australian and international researchers identify a new...
Blood test for chronic fatigue syndrome developed
The test addresses the need for a quick and reliable diagnostic for a complex,...




