Moderna vaccine approved for at-risk under-fives
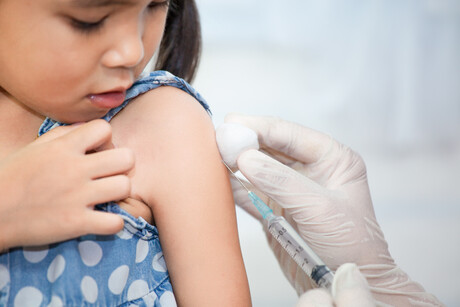
The Australian Government has accepted a recommendation from the Australian Technical Advisory Group on Immunisation (ATAGI) to make Moderna’s COVID-19 vaccine available to children aged six months to less than five years in certain at-risk population groups, with first doses set to begin on 5 September.
The ATAGI recommendation has been a long time coming, with the Therapeutic Goods Administration (TGA) having provisionally approved a paediatric dose of the Moderna vaccine for use in individuals aged six months to five years on 19 July. At this stage, ATAGI has chosen to recommend the vaccine only to those young children with severe immunocompromise, disability, and complex and/or multiple health conditions which increase the risk of severe COVID-19 — estimated to be around 70,000 individuals — stating that otherwise healthy young children have a very low likelihood of severe illness from COVID-19.
COVID-19 vaccination is already recommended for all children aged five years and above as per ATAGI’s previous advice. Previously, only the Pfizer vaccine was registered for use in 5-year-olds and Moderna was registered from six years of age; 5-year-old children may now receive either a Moderna or Pfizer vaccine (ideally the same brand should be used for all doses of a child’s primary vaccination schedule).
As with other age groups, the use of the Moderna vaccine in children from six months to five years should be administered as two doses (or three in the case of severe immunocompromise) at least four weeks apart (ATAGI recommends eight weeks). The paediatric vaccine is made in the same way as Moderna’s vaccines for older persons but contains a lower concentration of the active ingredient, at 25 µg in a 0.25 mL vial (versus 50 µg in a 0.25 mL vial for ages 6–11 years and 100 µg in a 0.5 mL vial for 12 years and older).
The government has secured 500,000 doses of the vaccine for this youngest age group, with initial supplies set to arrive in Australia later this week. Given the particular needs of this group of children and the small size of the cohort to be vaccinated, only a small proportion of vaccination sites will deliver this vaccine. These sites will be identified on the Vaccine Clinic Finder when bookings open later this month.
Please follow us and share on Twitter and Facebook. You can also subscribe for FREE to our weekly newsletters and bimonthly magazine.
AusBiotech partners with Tenmile
Designed to support Australia's homegrown life sciences innovation, AusBiotech has announced...
Australian CDC issues update in wake of Ebola outbreak
After the WHO determined the outbreak of Ebola in the DRC and Uganda to be a public health...
Australia announces $7.2m diphtheria outbreak response package
To respond to the biggest diphtheria outbreak on record, support has been announced for the NT...



