Drug transporters bring cancer medication to its destination
German researchers have developed an innovative technology designed to treat cancer more selectively and effectively, transforming proteins and antibodies into stable, highly functional drug transporters with which tumour cells can be detected and killed.
Classic chemotherapy for the treatment of cancer is based on toxic substances that are particularly effective for rapidly dividing cells. However, since healthy tissue also depends on cell division, treatment with chemotherapeutic substances is often accompanied by severe side effects — a dose sufficient to completely remove the tumour would in many cases be too toxic to administer to a diseased person.
With more modern approaches, it is now possible to transport active agents (drugs) in the body selectively to the site of action, for example, by linking a drug with an antibody that can differentiate cancer cells from healthy tissue through changes on the surface of the cell. Five such antibody-drug conjugates (ADCs) are already on the market; however, these ADCs lose a large part of their ‘toxic cargo’ en route to the cancer cell, causing the drugs to be released into the bloodstream and dangerous side effects to occur.
A stable link between drug and antibody would therefore be highly desirable, and was the focus of recent research from Leibniz-Forschungsinstitut für Molekulare Pharmakologie (FMP) and the Ludwig-Maximilians-Universität München (LMU). Writing in the journal Angewandte Chemie, the researchers presented two consecutive articles detailing the development of new methods and the application of these methods to selective drug transport.
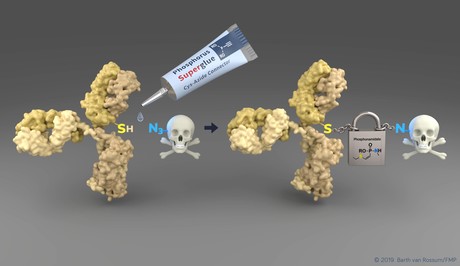
“We have developed an innovative technology that makes it possible to link native proteins and antibodies to complex molecules, such as fluorescent dyes or drugs, more easily and with better stability than ever before,” said Marc-André Kasper, a researcher at the FMP.
The researchers discovered the outstanding properties of unsaturated phosphorus (V) compounds and took advantage of those. These phosphonamidates connect a desired modification — for example, a cancer-fighting agent — exclusively to the amino acid cysteine in a protein or antibody. Since cysteine is a very rare natural occurring amino acid, the number of modifications per protein can be controlled quite effectively, which is essential for the construction of drug conjugates. In addition, phosphonamidates can easily be incorporated into complex chemical compounds.
“The greatest achievement of the new method, however, is that the resulting bond is also stable during blood circulation,” said Kasper. The ADCs that are on the market cannot achieve this.
To test the applicability on targeted drug delivery, the researchers compared their technology directly with an FDA-approved ADC, Adcetris. The medication was recreated as precisely as possible with the same antibody and active agent, the only difference being that the innovative phosphonamidate linkage was used.
When applied to blood serum, the researchers observed that their modified ADC lost significantly less active ingredient over a period of days. They also used the new technology in experiments with mice to combat Hodgkin’s lymphoma, where the preparation proved to be more effective than the conventional medication.
“From our results, we conclude that phosphonamidate-linked drug transporters can be administered in lower doses, and that side effects can be further reduced,” said FMP group leader Christian Hackenberger. “Thus, the technology has great potential to replace current methods in order to develop more effective and safer ADCs in the future.”
Please follow us and share on Twitter and Facebook. You can also subscribe for FREE to our weekly newsletters and bimonthly magazine.
AusBiotech partners with Tenmile
Designed to support Australia's homegrown life sciences innovation, AusBiotech has announced...
Australian CDC issues update in wake of Ebola outbreak
After the WHO determined the outbreak of Ebola in the DRC and Uganda to be a public health...
Australia announces $7.2m diphtheria outbreak response package
To respond to the biggest diphtheria outbreak on record, support has been announced for the NT...



